Translational Science Benefits
Clinical & medical benefits (Tools & products)
Definition
Apparatus, instruments, and materials for diagnostic, surgical, therapeutic, and scientific procedures. Equipment and supplies include scientific instruments used for basic discovery and clinical applications.
Rationale
Improvements in basic scientific instruments enhance the capacity of clinical researchers to make discoveries. Improvements in medical equipment translate scientific advances into improved patient outcomes by reducing instrument fatigue, improving recovery times, and reducing risk of injury.
Guidance
Advice from authors on searching for information:
This indicator may not be obvious to a researcher, as equipment and supplies may be designed, redesigned, or used in novel ways in support of a clinical and translational research project, but may not be the original project goal. Researcher interviews may be required to determine if new or redesigned equipment and supplies were used as a result of the project.
Resources & Data
Organizations, repositories, websites, and other sources where you can find more information:
Local
- Instrument Machine Shop. The Instrument Machine Shop offers design, fabrication, and repair services for parts, equipment, and scientific instruments. They can fabricate from sketches or formal blueprints and can fabricate original designs. The shop serves both Washington University and non-university customers.
- Office of Technology Management at Washington University (OTM). The OTM offers a Technology Search feature to search for all available Washington University technologies. For more information, consult the office at your institution that manages technology transfer and commercialization.
General
- Devices@FDA provides a search of cleared and approved medical devices. See the User Instructions for additional information.
- Online Commercial Vendors. Vendors such as Emergency Medical Products, Inc., AliMed, theEMSstore, Labcompare, etc. offer a wide range of medical supplies. Products can often be searched for by name, or browsed by use category, brand name, etc.
- U.S. Food and Drug Administration (FDA). Searches for “equipment” yield resources including qualification, validation, and maintenance of clinical and research equipment.
Data Limitations
Challenges you may encounter while searching for information:
Government websites such as FDA and CDC do not have sections specifically devoted to equipment or supplies, but do address this indicator in a wide variety of contexts.
Publications
Articles, books, and other publications in translational science using the benefit:
- Guidelines for pediatric equipment and supplies for emergency departments. Committee on Pediatric Equipment and Supplies for Emergency. Ann Emerg Med. 1998 Jan; 31(1): 54-57. doi: 10.1016/S0196-0644(98)70281-1.
- Hampson G. Practice nurse handbook, 5th ed. Oxford: Blackwell Publishing; 2008. Appendix 1, Examples of the Clinical Equipment Needed in the Nurses’ Rooms; p. 321-324. doi: 10.1002/9780470775110.
- Keay S, McCarthy JP, Carey-Smith BE. Medical equipment libraries: Implementation, experience and user satisfaction. J Med Eng Technol. 2015 Aug, 39(6) 354-362. doi: 10.3109/03091902.2015.1059507.
Case Studies

Transforming Tears to Smiles: Reducing Needle-based Procedural Pain in Children with Cancer Globally
The Global Comfort Promise initiative reduces needle pain and anxiety for children with cancer in low- and middle-income countries, supporting better adherence to treatment plans and ultimately improving health and survival outcomes
Clinical & medical benefits
Community & public health benefits
Economic benefits
Policy & legislative benefits
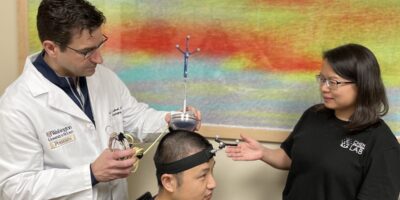
Detecting Brain Cancer Without a Knife
Facilitating brain cancer diagnosis with an innovative and portable ultrasound device
Clinical & medical benefits
Community & public health benefits
Economic benefits